Chem Equations Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 10 clues. Answers range from 6 to 18 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
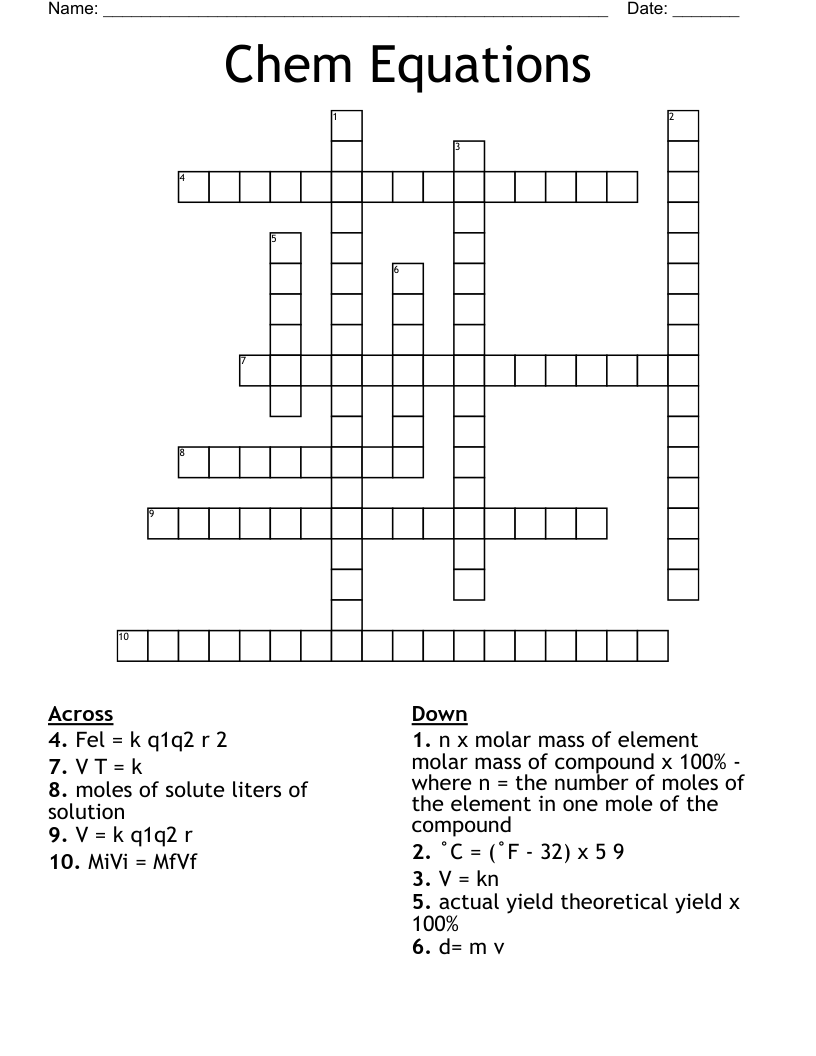
d= m v
˚C = (˚F - 32) x 5 9
n x molar mass of element molar mass of compound x 100% - where n = the number of moles of the element in one mole of the compound
moles of solute liters of solution
MiVi = MfVf
V T = k
actual yield theoretical yield x 100%
V = kn
Fel = k q1q2 r 2
V = k q1q2 r
Customize
Add, edit, delete clues, and customize this puzzle. Print copies for an entire class.