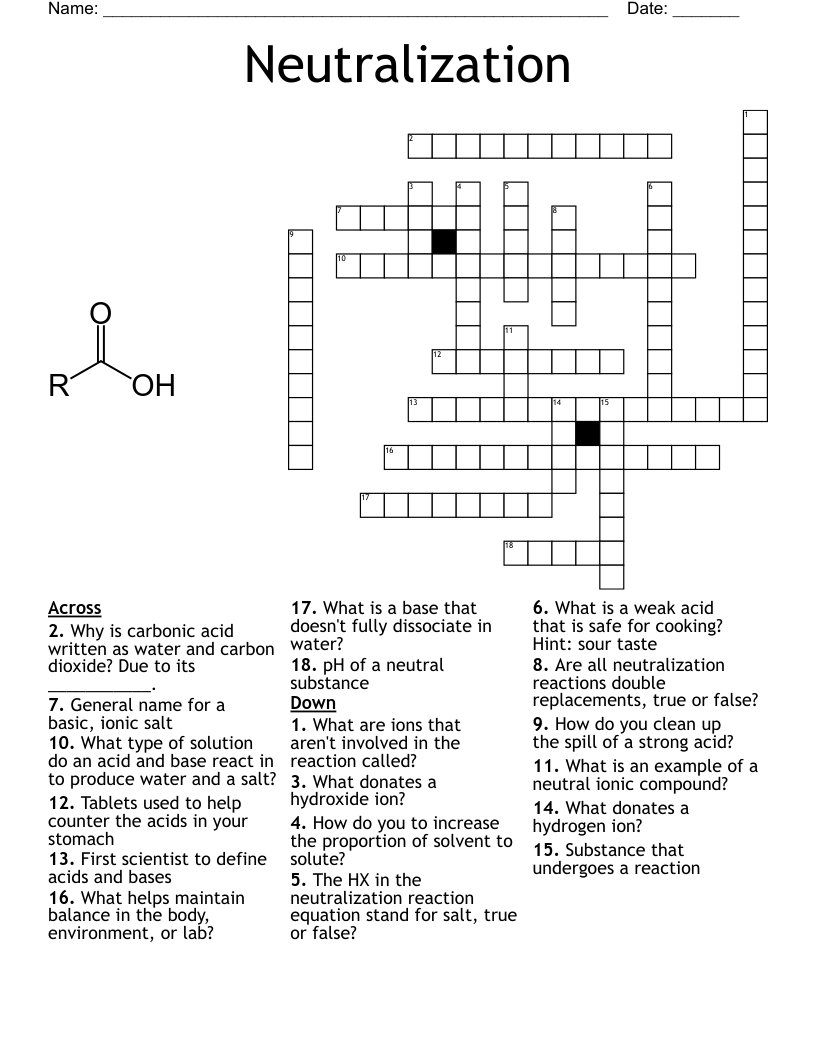
Chemistry Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 21 clues. Answers range from 2 to 15 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
Having a pH of less than 7, and turns litmus red; typically, a corrosive or sour-tasting liquid of this kind.
Slippery to the touch, can taste bitter if an alkali, changes the color of indicators, turn red litmus paper blue
Mixture in which one substance of microscopically dispersed insoluble particles is suspended throughout another substance.
Refers to the amount of a substance per defined space. ... However, the solute ___________ may also be expressed in moles or units of volume.
General process in which molecules (or ionic compounds such as salts, or complexes) separate or split into smaller particles such as atoms, ions or radicals
The ion H3O+, consisting of a protonated water molecule and present in all aqueous acids
Any substance that gives a visible sign, usually by a color change, of the presence or absence of a threshold concentration of a chemical
Any process by which electrically neutral atoms or molecules are converted to electrically charged atoms or molecules
A concentration unit, defined to be the number of moles of solute divided by the number of liters of solution.
a chemical solution which is neither acidic nor basic.
Measure of hydrogen ion concentration, a measure of the acidity or alkalinity of a solution.
The point at which a solution of a substance can dissolve no more of that substance.
A chemical property referring to the ability for a given substance, the solute, to dissolve in a solvent.
A substance dissolved in another substance
a liquid mixture in which the minor component (the solute) is uniformly distributed within the major component (the solvent).
Component of a solution that is present in the greatest amount. It is the substance in which the solute is dissolved.
Any acid that ionizes completely in solutions.
A state of a solution that contains more of the dissolved material than could be dissolved by the solvent under normal circumstances.
Mixture whose size of particles are large enough to settle out
Slow addition of one solution of a known concentration (called a titrant) to a known volume of another solution of unknown concentration until the reaction reaches neutralization
An acid that is partially dissociated into its ions in an aqueous solution or water.