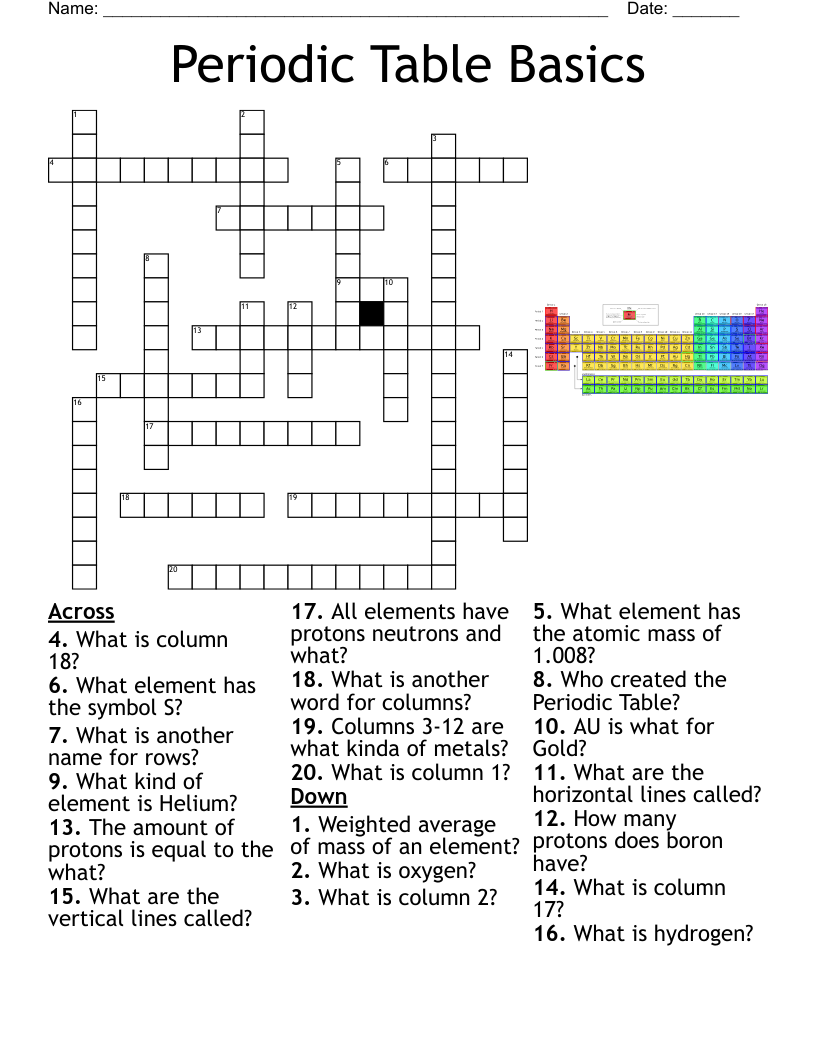
Chemistry Review Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 30 clues. Answers range from 3 to 18 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
A change in matter that produces new substances with new properties. (2words)
New substances produced in a chemical reaction.
Substances that react together in a chemical reaction.
A system for organizing elements into columns and rows based on their properties. (2words)
Rows in the periodic table.
Columns in the periodic table.
A group of letters and subscript numbers that represent the make-up of a chemical compound. (2words)
An atom that has an electrical charge, either positive or negative.
A compound that forms between a metal and a non-metal after electrons are transferred.
A compound that forms between non-metals as they share electrons.
A subatomic particle found in the nucleus of an atom. It has no charge.
A positively charged subatomic particle found in the nucleus of the atom.
A negatively charged subatomic particle that orbits the nucleus of an atom.
Refers to either the outer orbital of an atom or the electrons found in the outer shell.
A reaction in which two or more reactants combine to produce a new product.
A reaction in which one compound breaks down into two or more simpler compounds.
A reaction in which one element takes the place of another element in a compound. (2words)
A reaction in which the metal ions of two different compounds exchange places. (2words)
The smallest unit of an element.
A compound that tastes sour, corrodes metal and tissue, and turns blue litmus paper red.
A substance that changes colour when added to an acid or a base.
A compound that tastes bitter, feels slippery, corrodes tissue, and turns red litmus paper blue.
The number in front of a product or reactant in a balanced chemical equation.
Ability to conduct (transfer) electricity.
Law describing the fact that the total mass and number of atoms is the same before and after a reaction. (3words)
A reaction between an acid and a base that produces salt and water.
Elements on the left side of the periodic table. They are malleable, shiny, conductive, and solid at room temperature.
Elements on the right side of the periodic table. They are not malleable, not conductive, and exist in a number of states at room temperature.
A pure substance that cannot be broken down.
A pure substance that contains two or more different elements.