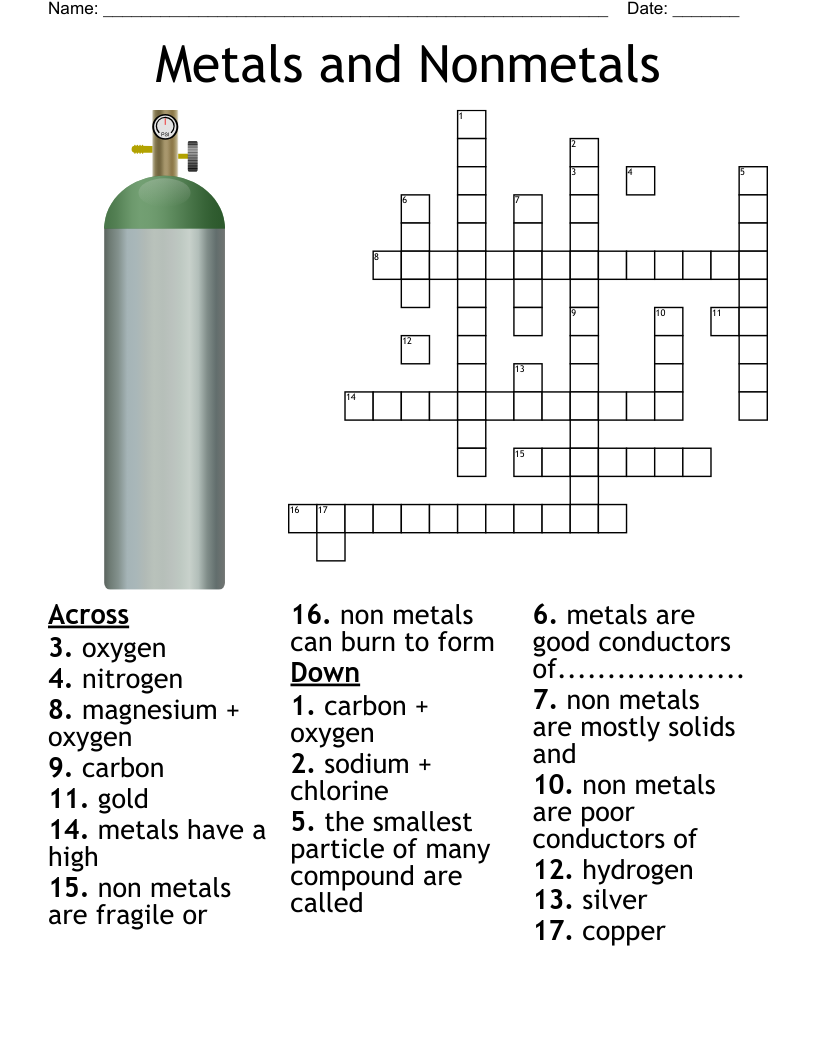
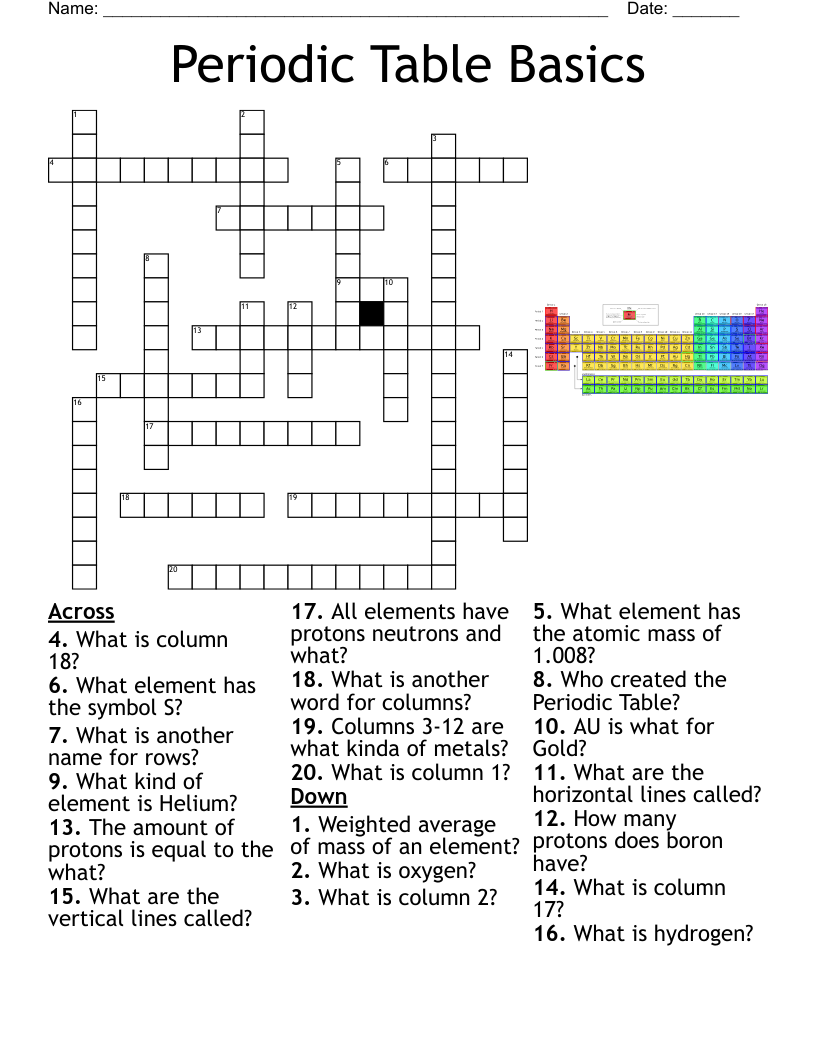
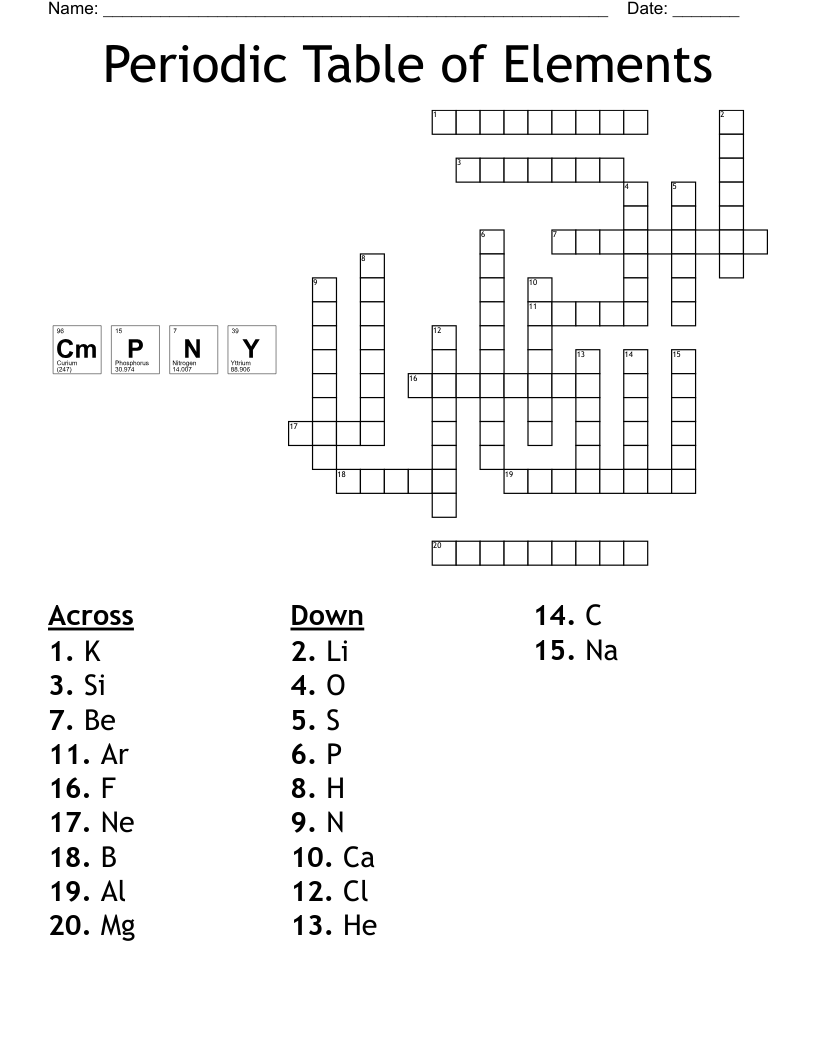
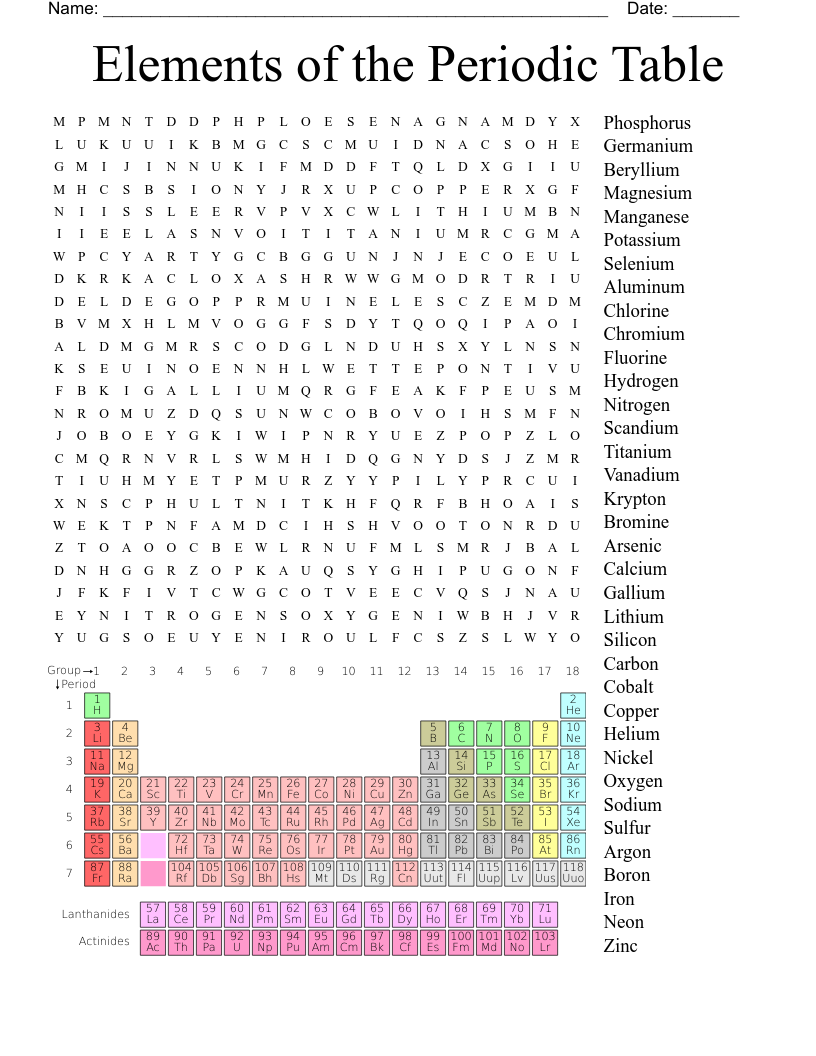
Periodic Table Crossword
This printable crossword puzzle on the topic of Elements & Periodic Table has 25 clues. Answers range from 3 to 19 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
Group 3 of the periodic table. These 15 metals, whose atomic numbers range from 89-103, are radioactive.
Group 1 of the periodic table. These 6 metals are very reactive and get more reactive as you go down.
Group 2 of the periodic table. These 6 metals share similar characteristics with group 1.
The smallest particle that has the properties of an element
The total number of protons and neutrons in an atoms nucleus
The unit of measure used to measure the mass on an atomic level
The number of protons and electrons in the nucleus of an atom
Number of groups in the periodic table
A substance that is made entirely from one type of atom
Abbreviation for an element's name
Vertical columns of elements in the periodic table
Group 17 of the periodic table. These 6 non-metals are toxic and are located to the left of the noble gases.
An atom or a group of atoms that has lost or gained one or more electrons and therefore has a net electric charge
Any atoms having the same number of protons but a different number of neutrons
Group 3 and Period 6 of the periodic table. These 15 metals, whose atomic numbers range from 57-71, are located above the actinides.
An element that has properties of both a metal and nonmetal
The elements that are good conductors of heat and electricity
Group 18 of the periodic table. These 6 non-metals are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity.
The elements that are usually poor conductors of heat and electricity
The properties of elements tend to repeat in a regular pattern when elements are arranged in order of increasing atomic number
The arrangement of elements into families and rows based on similar features and characteristics
Horizontal rows of elements in the periodic table
Number of periods in the periodic table
Groups 3-12 of the periodic table. These metals serve as a bridge- or transition between the two sides of the table.
An electron in the outermost energy level of an atom