Unit 3 Crossword Puzzle
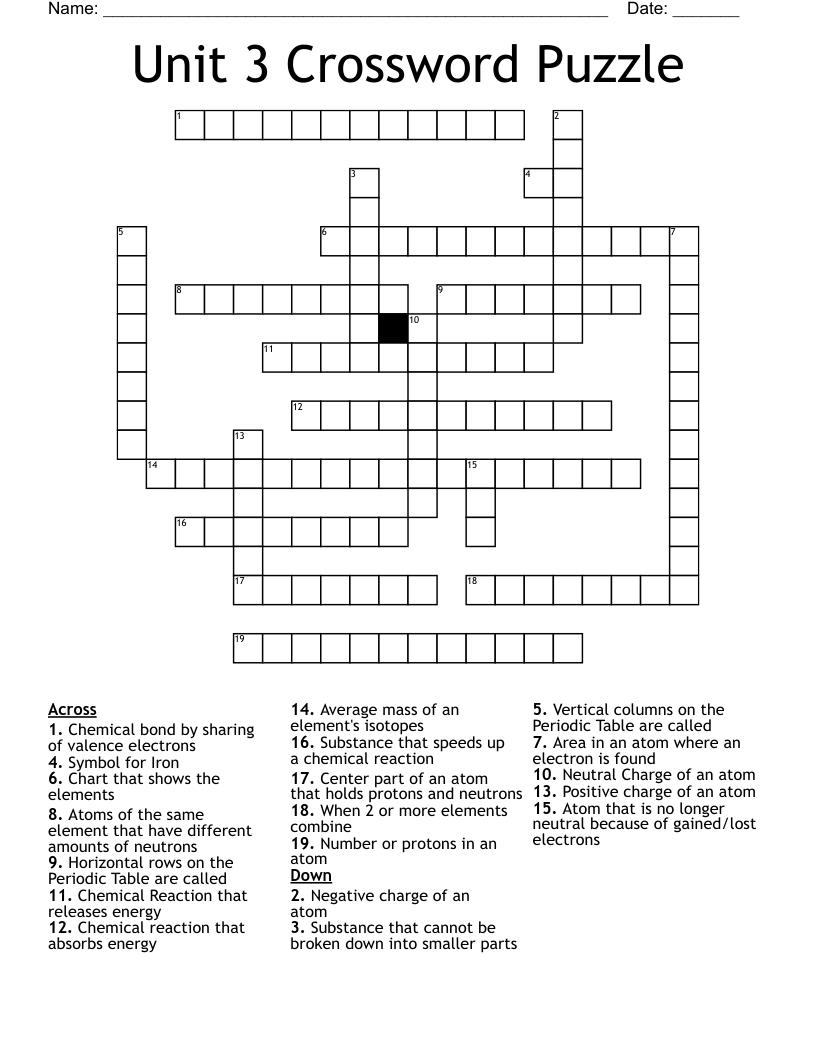
This printable crossword puzzle on the topic of Elements & Periodic Table has 19 clues. Answers range from 2 to 17 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
Negative charge of an atom
Center part of an atom that holds protons and neutrons
Positive charge of an atom
Neutral Charge of an atom
Area in an atom where an electron is found
Substance that cannot be broken down into smaller parts
Number or protons in an atom
Atoms of the same element that have different amounts of neutrons
Average mass of an element's isotopes
Chart that shows the elements
When 2 or more elements combine
Atom that is no longer neutral because of gained/lost electrons
Chemical bond by sharing of valence electrons
Chemical Reaction that releases energy
Chemical reaction that absorbs energy
Substance that speeds up a chemical reaction
Symbol for Iron
Vertical columns on the Periodic Table are called
Horizontal rows on the Periodic Table are called