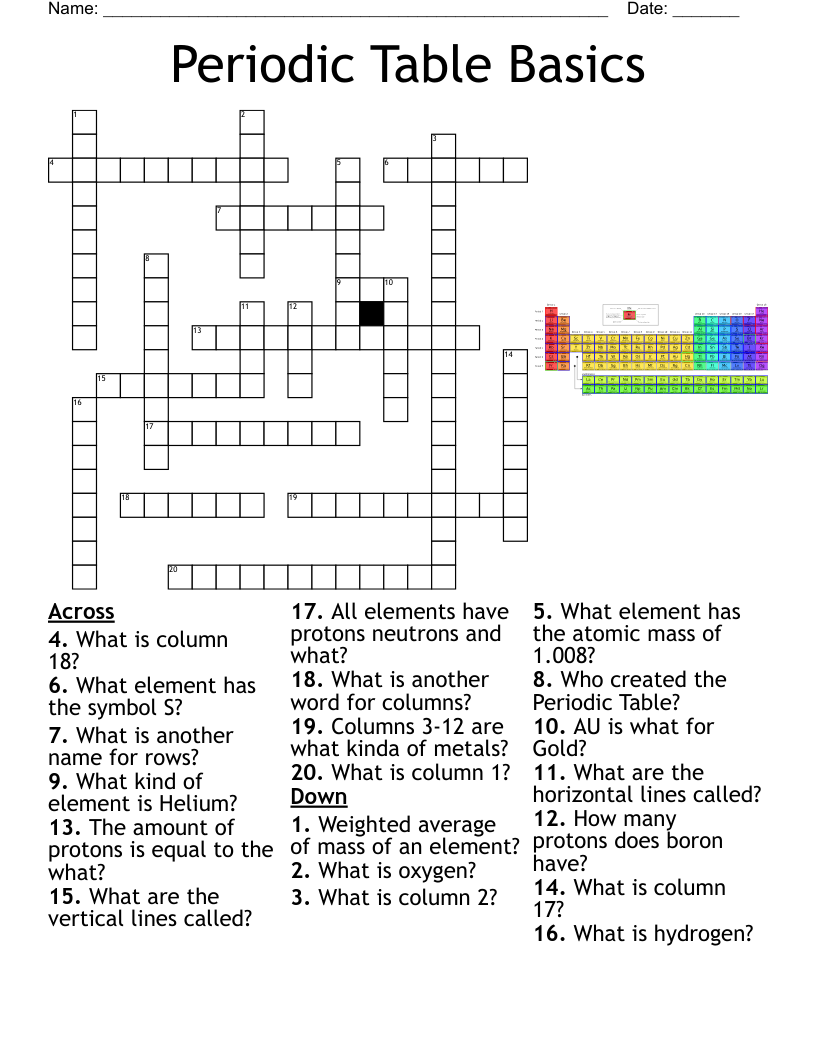
Periodic Table Vocabulary Crossword
This printable crossword puzzle on the topic of Elements & Periodic Table has 20 clues. Answers range from 4 to 19 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
a substance that cannot be broken down into a simpler substance by ordinary chemical means
the smallest unit of an element that has all of the properties of the element; basic building block of matter.
the number of protons and neutrons in the nucleus of one atom of the element.
the number of protons contained in each nucleus of its atoms of the element.
a horizontal row (left to right) in the periodic table.
a vertical column (up and down) on the periodic table.
a chart that organizes information about all of the known elements according to their atomic number.
describes how likely an element is to form bonds with other elements.
an element or substance that conducts heat and electricity, is malleable and ductile and has low ionization energy and low electronegativity values. Metals
an element that does not conduct electricity or heat and is usually a gas at room temperature. Nonmetals are brittle, have high ionization energies and high electronegativity values. Nonmetals tend to gain electrons to form anions
group 1 metals on the periodic table that contain 1 valence electron and lose their valence electrons the most easily, making them the most reactive metals.
group 1 metals on the periodic table that contain 2 valence electrons and are the second most reactive metals.
group 3-12 on the periodic table. They have varying valence electrons and do not follow the normal trends of the other metals. They form brightly colored compounds and ions in solution.
group 17 nonmetals on the periodic table that contain 7 valence electrons. They only need to gain 1 valence electron to have a stable octet. They gain valence electrons the most readily, making them the most reactive nonmetals.
group 18 elements on the periodic table that contain 8 valence electrons (He has 2) and a full valence shell making them very stable and inert.
an element that has some properties of a metal and some properties of a nonmetal. The metalloids are found on the boron staircase, there are 7 metalloids: B, Si, Ge, As, Sb, Te, and Po.
elements and/or compounds that when put together are unable to react chemically. The noble gases (group 18) elements are inert because of a full valence shell.
the charge of an atoms nucleus resulting from its number of protons
the distance between the nucleus of an atom and it's outermost energy level
the attraction a nucleus has resulting from its number of protons