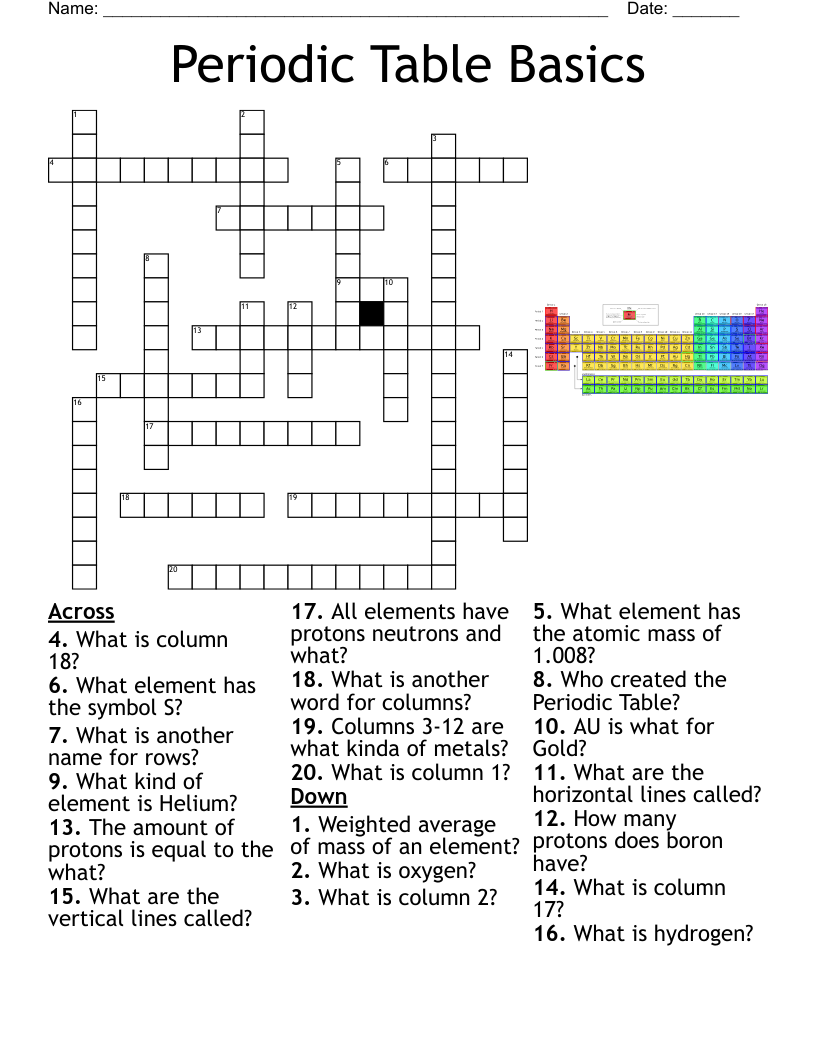
Chemistry Vocabulary Puzzle 2 Crossword
This printable crossword puzzle on the topic of Physics has 31 clues. Answers range from 4 to 22 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
a negatively charged particle; found in the electron cloud.
a particle with a neutral charge; found in the nucleus.
the center of that atom that contains the protons and neutrons.
identify an atom, positive charge; found in the nucleus
he number of protons and neutrons in an element.
the same as the number of protons
two or more forms of the same elements with the same amount of protons but different amounts of neutrons (different amu). Sometimes it is the radioactive form of the element.
the amount of protons and neutrons in an element (amu)
a helium particle emitted by some radioactive elements, +2 charge.
ionizing radiation with alpha particles emitted during radioactive decay.
radioactive decay where beta particles are emitted.
a particle with a charge of -1.
smallest wavelength with the most amount of energy. High energy photons, no charge.
represents the reactants and products of radioactive decay.
the change in identity of an atomic nucleus when it is bombarded with an energy particle
radioactive substances or the emissions emitted
emissions of energy through waves or particles
when an unstable atom loses energy by radiation.
the length of an electron beam transmitted for a substance.
isotopes with unstable nuclei.
a form of ionizing radiation, energy is deposited into matter.
the stability of elements determined by the ratio if the number of neutrons to the number of protons in the nucleus.
the time requires for half of the original unstable isotope to decay to another atom
a proton or neutron.
a decay through a sequence of alpha and beta decays until a stable element is reached
determines the age of an object by measuring the amount of carbon radioisotopes remaining
one of the four fundamental forces in nature
the changing from one element to another by radioactive deca/bombardment.
difference in mass between a nucleus and its component nucleus.
when a nucleus splits due to the bombardment of energy, produces large amounts of energy compared to chemical reactions
a nuclear reaction where a nucleus with a low atomic number fuses to form a heavier nucleus