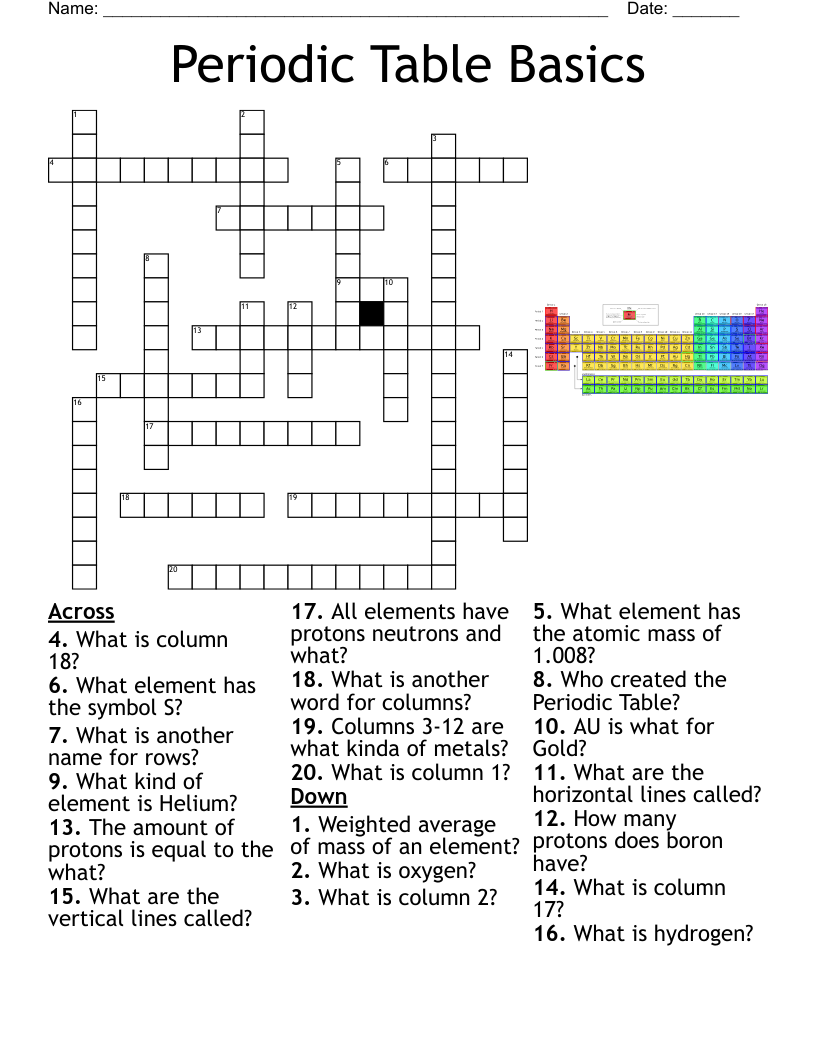
periodic table project Crossword
This printable crossword puzzle on the topic of Elements & Periodic Table has 29 clues. Answers range from 4 to 18 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
the basic unit of a chemical element.
a table of the chemical elements arranged in order of atomic number, usually in rows, so that elements with similar atomic structure
is measurable, whose value describes a state of a physical system
. a property or characteristic of a substance that is observed during a reaction in which the chemical composition or identity of the substance is changed:
is a column of elements in the periodic table of the chemical elements
s is the small, dense region consisting of protons and neutrons at the center of an atom,
is a measure of the size of its atoms,
a stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids.
a stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron, but of opposite sign.
a subatomic particle of about the same mass as a proton but without an electric charge,
the central and most important part of an object, movement, or group, forming the basis for its activity and growth.
a thing that is composed of two or more separate elements; a mixture
a single substance
a solid material that is typically hard, shiny, malleable, fusible, and ductile, with good electrical and thermal conductivity
an element whose properties are intermediate between those of metals and solid nonmetals.
the theory that matter consists of indivisible particles called atom
the electrode of an electrochemical cell at which reduction occurs
. It is used to describe where electrons are when they go around the nucleus of an atom.
New Zealand-born British physicist who classified radiation into alpha, beta, and gamma types and discovered the atomic nucleus.
He formulated the Periodic Law, created a farsighted version of the periodic table of elements,
He formulated the Periodic Law, created a farsighted version of the periodic table of elements,
the number of protons in the nucleus of an atom, which determines the chemical properties of an element and its place in the periodic table.
each of two or more forms of the same element that contain equal numbers of protons but different numbers of neutrons in their nuclei
the mass of an atom of a chemical element expressed in atomic mass units.
is a measure of the tendency of an atom to attract a bonding pair of electrons
is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons to form ions, often in conjunction with other chemical changes.
any of the elements lithium, sodium, potassium, rubidium, cesium, and francium, occupying Group IA (1) of the periodic table.
any of the elements fluorine, chlorine, bromine, iodine, and astatine, occupying group VIIA (17) of the periodic table.
any of the gaseous elements helium, neon, argon, krypton, xenon, and radon, occupying Group 0 (18) of the periodic table.