Atom image from ClipSafari
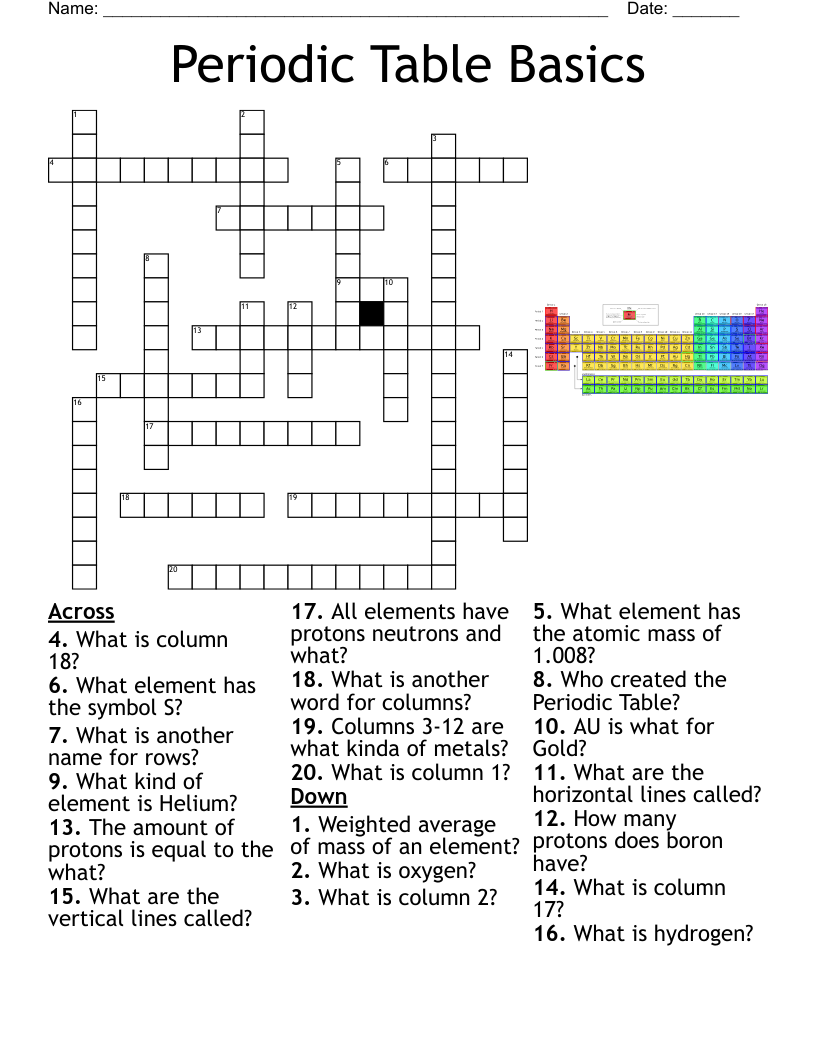
Atomic Structure Crossword Puzzle
This printable crossword puzzle on the topic of Elements & Periodic Table has 15 clues. Answers range from 4 to 20 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
The smallest unit of an element that maintains the properties of that element.
A subatomic particle that has no change and that is found in the nucleus of an atom.
A region around the nucleus of an atom whose electrons are likely to be found.
A unit of mass that describes the mass of an atom or molecule.
An atom's central region, which is made up of protons and neutrons.
An atom that has the same number of protons/atomic number as other atoms of the same element do but that has a different number of neutrons/atomic mass.
The mass of an atomn expressed in atomic mass units.
A subatomic particle that has a positive charge and that is found in the nucleus of an atom.
A subatomic particle that has a negative charge.
The sum of the number of protons and neutrons in the nucleus of an atom.
The number of protons in the nucleus of an atom; the atom's number is the same for all atoms of an element.
Plays in radioactive atoms that changes a neutron to a proton or electron.
A type of physical that occurs between electrically charged particles.
The force that holds particles together in the atomic nucleus.
A force that attracts any objects with mass.
Atom image from ClipSafari