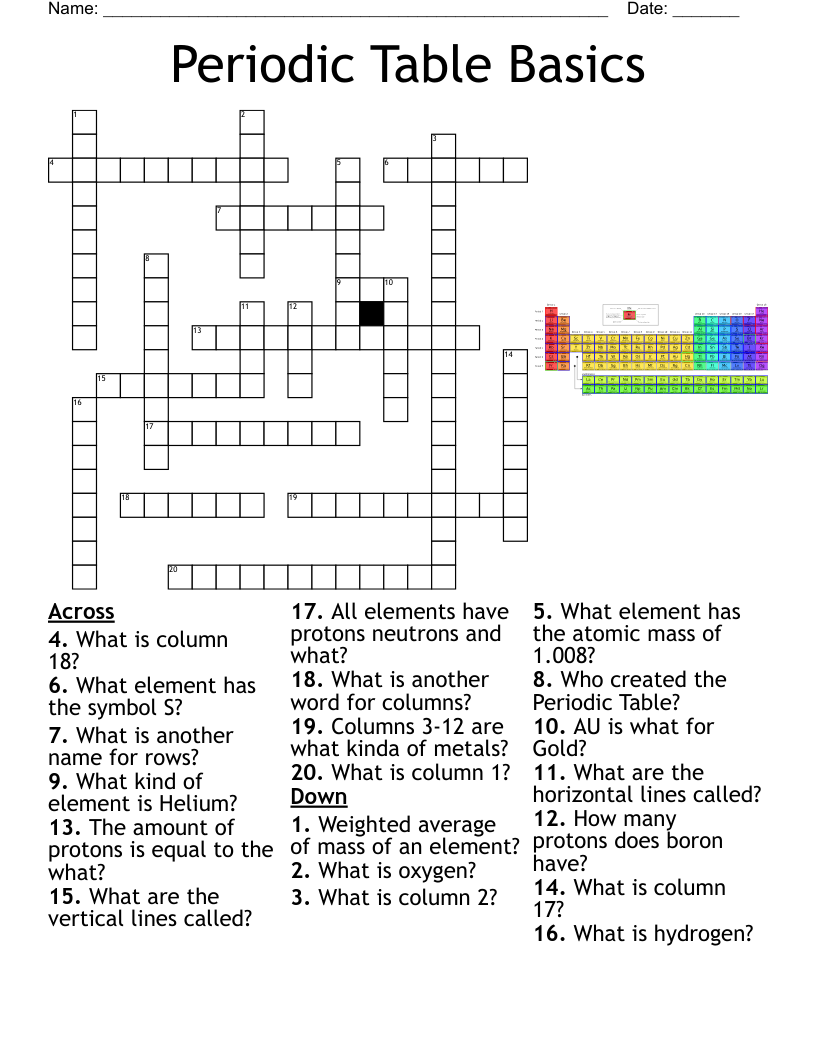
Atoms and the Periodic Table Vocabulary Crossword
This printable crossword puzzle on the topic of Elements & Periodic Table has 22 clues. Answers range from 4 to 18 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
a pure substance that cannot be separated or broken down into simpler substances by physical or chemical means
a pure substance composed of two or more elements that are chemically combined
elements that are shiny and are good conductors of thermal energy and electric current; most of them are malleable and ductile
elements that are dull and are poor conductors of thermal energy and electric current
elements that have two properties of both metals and nonmetals; sometimes referred to as semiconductors
the smallest particle into which an element can be divided and still be the same substance
the negatively charged particles found in all atoms; they are involved in the formation of chemical bonds
the tiny, extremely dense, positively charged region in the center of an atom; made up of protons and neutrons
the regions inside an atom where electrons are likely to be found
the positively charged particles of the nucleus; the number of this particle in a nucleus is the atomic number that determines the identity of a substance
the particles of the nucleus that have no charge
the number of protons in the nucleus of an atom
the weighted average of the masses of all he naturally occurring isotopes of an element
atom that have the same number of protons, but have different numbers of neutrons
the sum of the protons and neutrons in an atom
a column of elements on the periodic table
a horizontal row of elements on the periodic table
the law that states that the chemical and physical properties of elements are periodic functions of their atomic numbers
the elements in Group 1 of the periodic table; they are the most reactive metals, their atoms have one electron in their outer level
the elements in Group 2 of the periodic table; they are reactive metals, but are less reactive than alkali metals; their atoms have two electrons in their outer level
the elements in Group 17 of the periodic table; they are very reactive nonmetals, and their atoms have seven electrons in their outer level
the unreactive elements in Group 18 of the periodic table; their atoms have eight electrons in their outer level