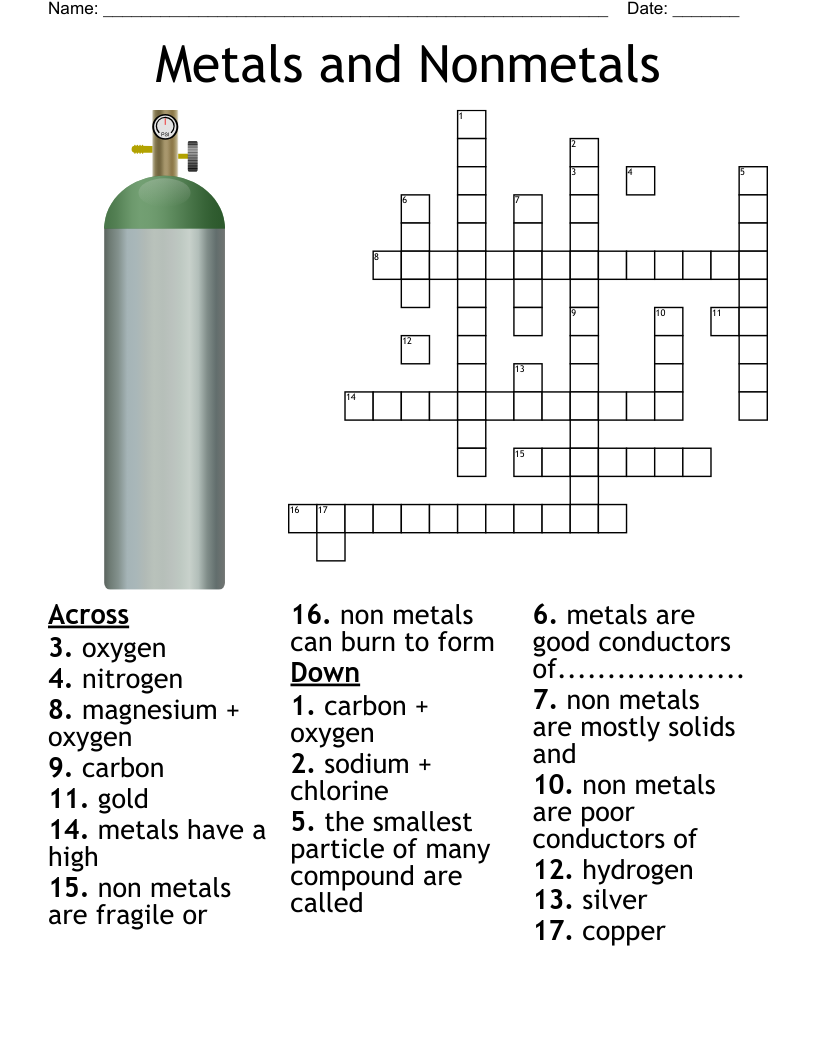
Metal Structures Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 15 clues. Answers range from 4 to 13 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
The particles in a metal are held together by strong _________________________
It takes a lot of _______________ to separate the particles.
That is why they have high _________________ points and boiling points.
Solid metals are _________________.
This means that the particles are close together and in a ____________ arrangement.
Metals have loose electrons in the outer shells which form a 'sea' of detached negative charge around the close-packed _____________________.
There are strong ___________________ forces holding the particles together.
All metals are _________________, meaning they can be hammered into shape.
The advantage of malleability is that metals have many ________, eg for making jewellery.
The elements in the centre of the periodic table, between groups 2 and 3, are called the ____________ metals.
Most of the commonly used metals are there, including iron, _________, silver and gold.
Transition metals are good ________________ of heat and electricity
They are less reactive than ______________ metals such as sodium
Transition metals have __________ melting points - but mercury is a liquid at room temperature
Transition metals have high _______________