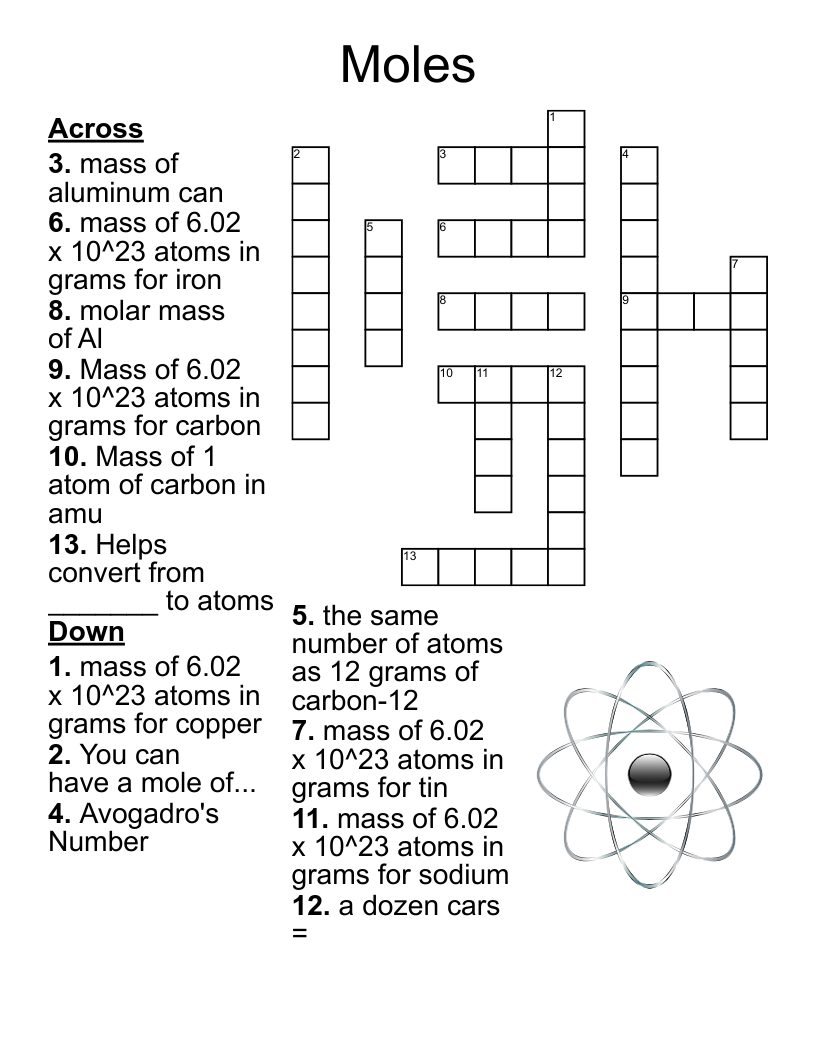
Chemistry Chapter 13 Crossword Puzzle
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 18 clues. Answers range from 7 to 27 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
States that the enthalpy change of reaction = the sum of the enthalpy changes from each step of the process.
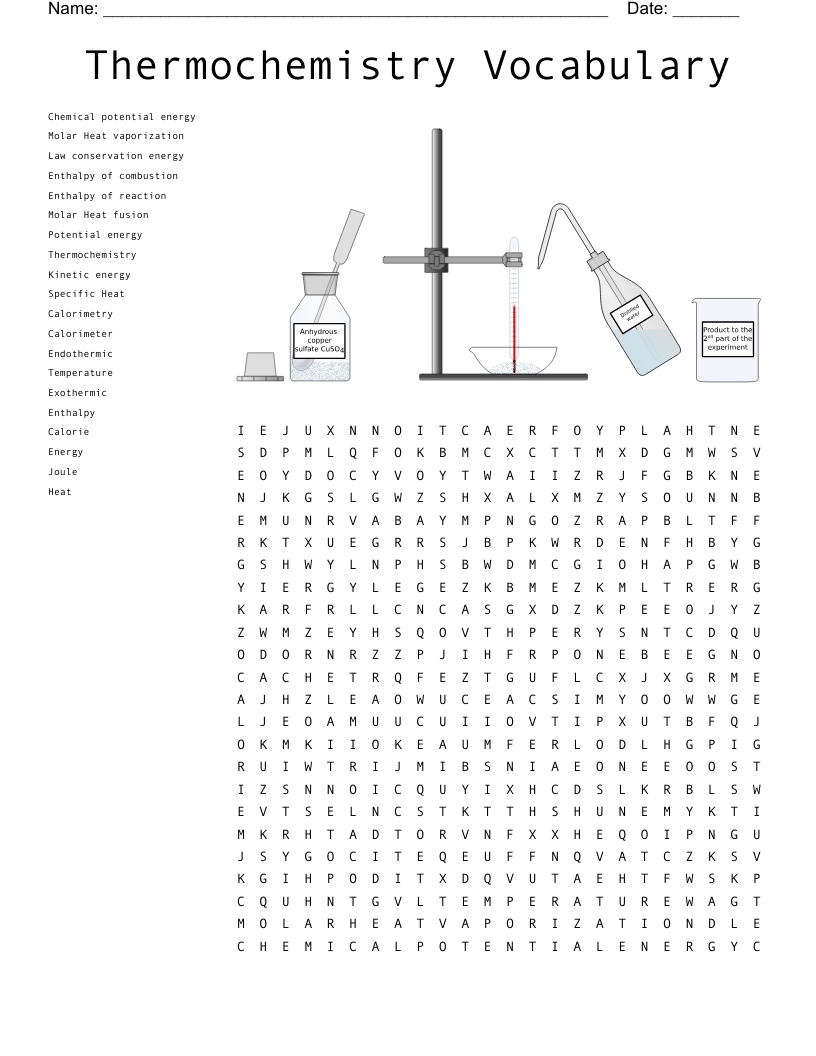
The amount of heat required to raise the temperature of 1g of the substance by 1°C
The type of heat that is sensed by a change in temperature.
25°C (298 K) and 1 atm of pressure
Delta(G)=Delta(H)-(T)Delta(S)
Measure of randomness or lack of orderliness in a system.
Change of enthalpy that occurs during a reaction.
The type of heat in which no temperature is observed; produces a phase change.
Instrument in which a thermometer detects temperature change that occurs during a chemical reaction.
The branch of science that studies the transfer of energy during a chemical reaction or phase change.
The heat content of a system at a constant pressure; helps quantify change in energy.
The quantity of heat required to melt one mole of a solid to a liquid with no temperature change.
The amount of heat required to convert a mole of liquid at its boiling point to its vapor at the same temperature.
Chemical equation that shows the physical state of each substance
The change in enthalpy that occurs when one mole of the compound is formed from its elements.
Enthalpy change for the reaction that produces 1 mole of a compound in its standard state from its elements in their standard states.
Energy is released as heat by the complete burning of one mole of a substance at standard conditions.
J. Willard Gibbs. The change in enthalpy and changes in entropy into a single term.