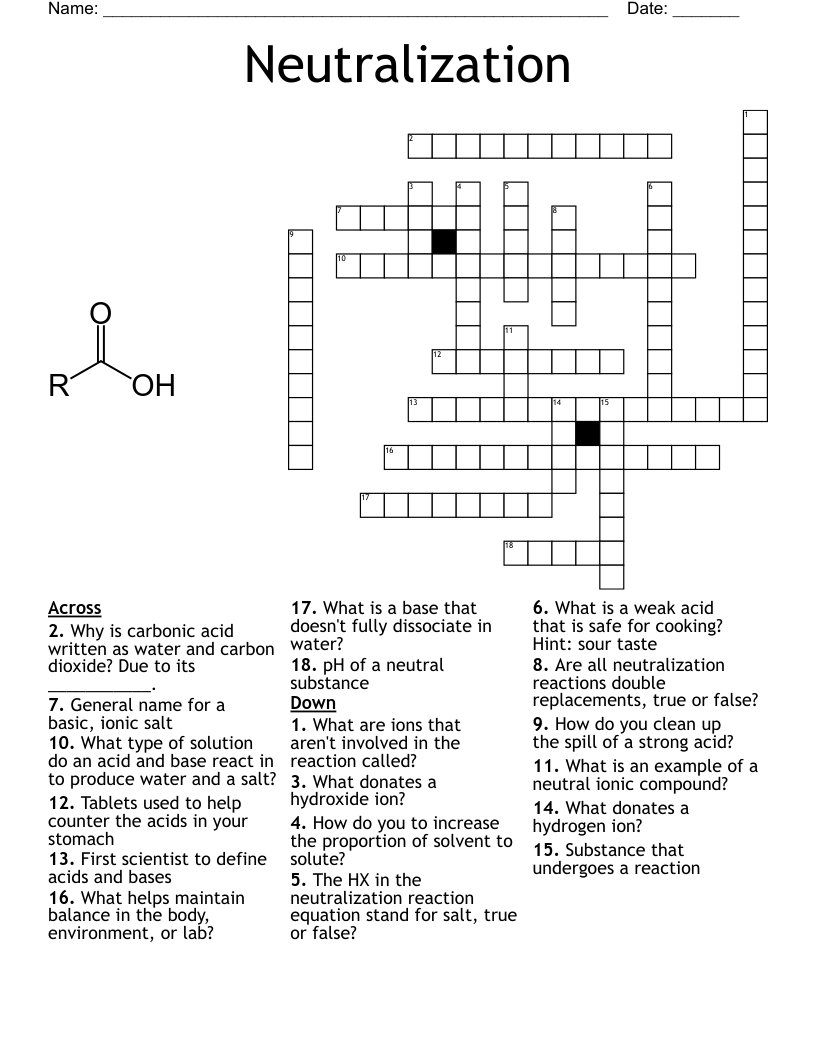
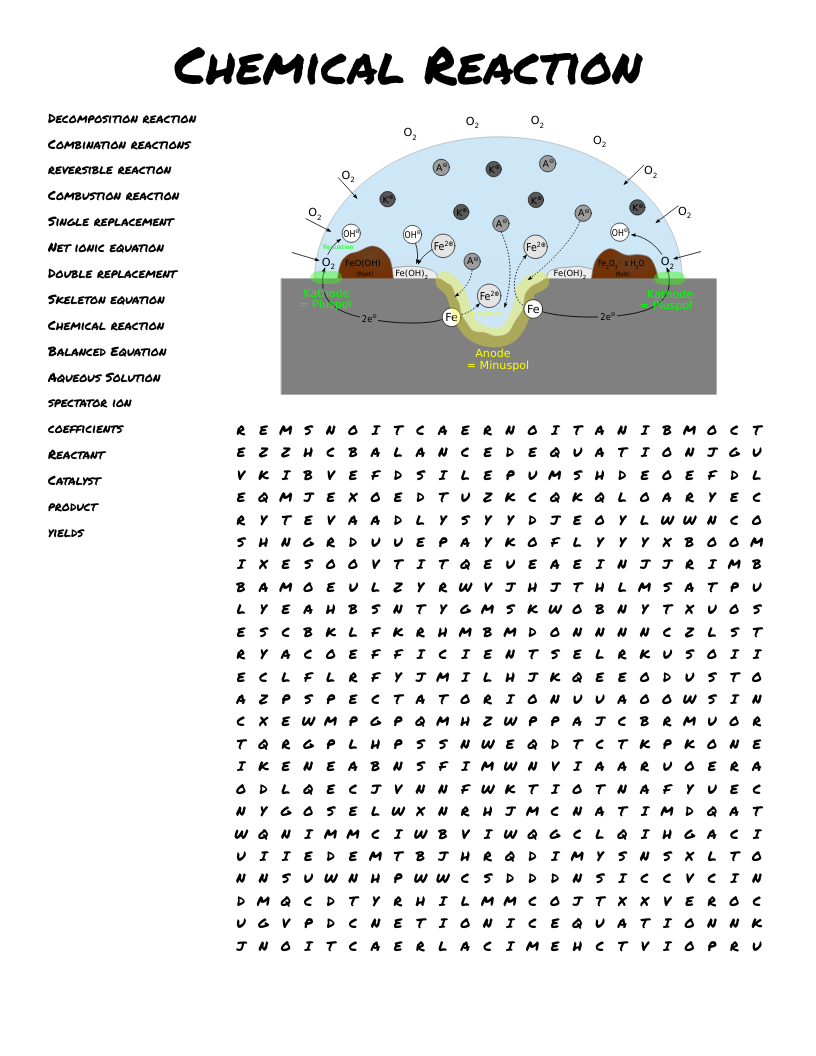
Chemical Kinematics Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 25 clues. Answers range from 7 to 29 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
the minimum energy required to initiate a chemical reaction
elementary reaction in which a single molecule is involved
rate at a particular moment of a reaction: determined from the slope of the curve of a graph at a particular point
the binding of molecules to a surface; usually the initial step in heterogeneous catalysis
elementary reaction involving the collision of two reactant molecules
the constant A in the Arrhenius equation that does not change as temperature is varied and is related to the frequency of collision and the probability that the collisions are favorably oriented for the reaction
the constant k in the rate law that changes with temperature and therefore determines how temperature affects rate
physical state of reactants, concentration of reactants, temperature, presence of catalyst
an equation that shows how the rate depends on the concentration of reactants
a substance that is neither a reactant nor product within the final net equation because it is formed in one elementary step and consumed in the next, having no overall effect on the multistep mechanism
the power to which the concentration of a reactant is raised in a rate law; determined experimentally or using the coefficients of reactants within a balanced equation
reaction whose rate depends on the concentration of a single reactant raised to the first power
a catalyst that exists in a different phase from the reactant molecules
characteristic determined by the number of molecules that participate as reactants in an elementary reaction
a catalyst that is present in the same phase as the reacting molecules
equation that incorporates the fraction of molecules possessing an energy Ea or greater, the number of collisions per second, and the fraction of collisions that have the appropriate orientation
model based on the kinetic-molecular theory that accounts for both the effects of concentration of reactants and temperature on rates at the molecular level
describes the process by which a reaction occurs
the particular arrangement of atoms at the top of the barrier, or the energy necessary to force through the relatively unstable intermidiate state of the final product
in the rate law, reaction rate depends on the concentration of the reactants not their volumes
net change represented by a balanced equation that consists of a sequence of elementary steps
since the overall reaction rate can not exceed the rate of the slowest elementary step in its mechanism, this slow step limits the overall reaction
does not affect rate in any way
the speed of a chemical reaction
elementary reaction involving the simultaneos collision of three molecules