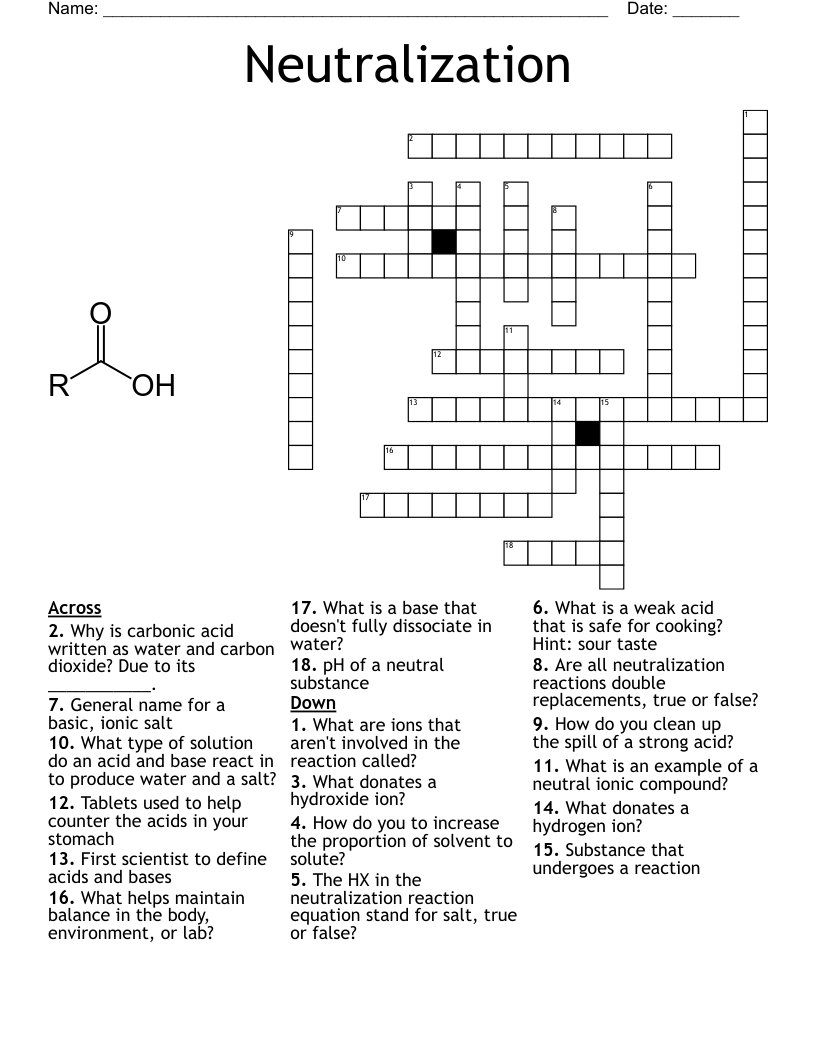
Chemical Kinetics and Chemical Equilibrium Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 15 clues. Answers range from 4 to 16 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
For Kc=[A][B]^2, if A is halfed and B is doubled, the value of Kc _____
Properties, concentration, temperature, and catalysts are examples of factors that affect _____ reactions.
How the exponents in a rate law equation are determined
This acts as a intermediary between the reactants and products in a reaction
Pure solids and ____ are left out of an equilibrium constant expression.
For the equation N2(g) + 2O2 (g) --> 2NO2(g), the concentration of NO2 is ____.
This happens to an enzyme if the temperature goes above or below the temperature range
This prevents a substrate from the action of an enzyme
The reaction A + 2B --> C is a _____ order reaction.
The unit for k in a first order reaction
For the reaction NH4Cl (aq) --> NH3(g) + HCl(g), the equation will shift ___ when pressure is increased.
"p" is known as this in the Arrhenius equation
This speeds up a reaction without being consumed
When the container volume is increased, the system will shift to ______ its volume.
This is constant in zero order reactions