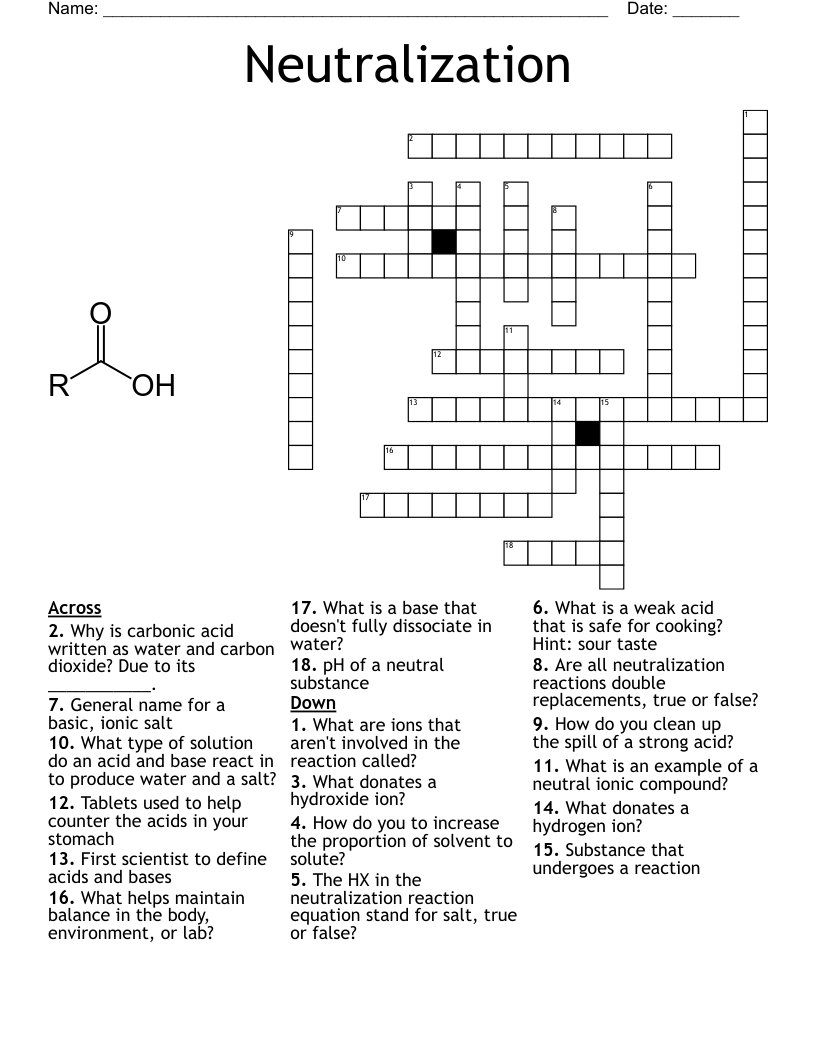
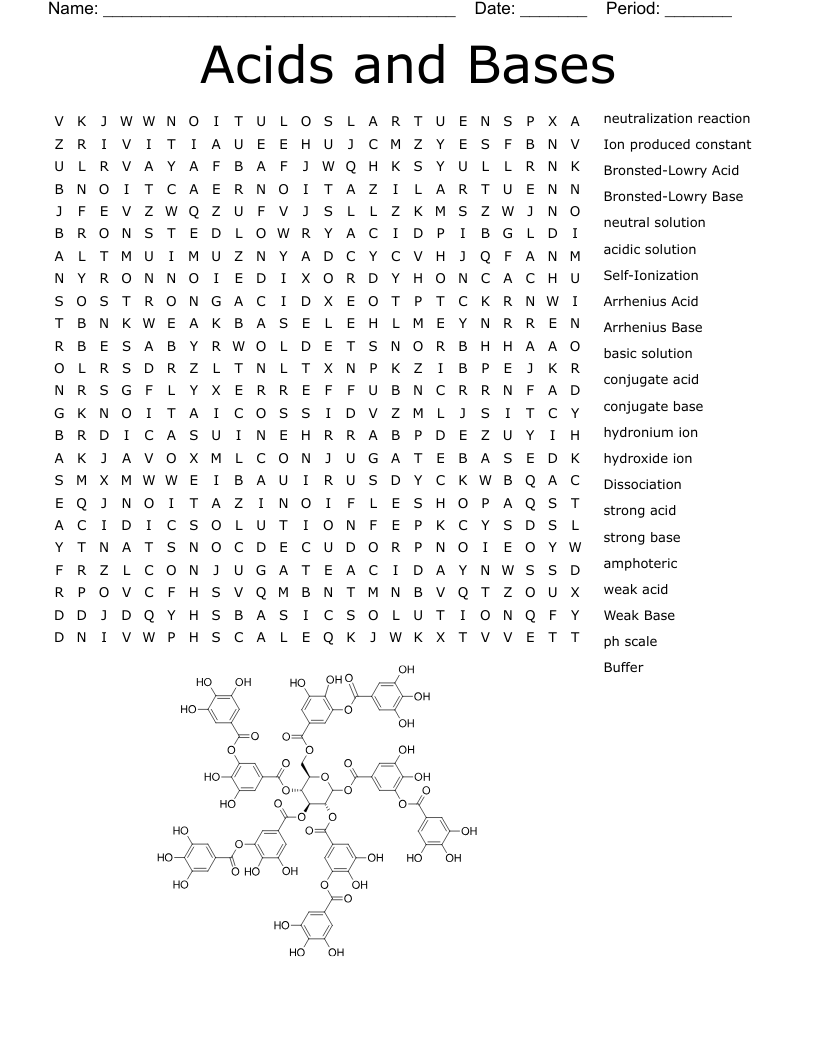
acids and bases 1 Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 31 clues. Answers range from 2 to 22 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
Acids that ionize completely in solution.
Acids that only ionize partially in solution.
Bases that dissociate entirely into metal ions and hydroxide (OH-) ions in aqueous solution (Arrhenius base).
Bases that ionize only partially in dilute aqueous solution to form the conjugate acid and hydroxide ions.
A measure of the strength of an acid or base solution which is based on the amount of H+ ion
A measure of the strength of an acid or base solution which is based on the amount of OH- ion.
Have pH < 7
Have pH = 7
Have pH > 7
HCl HBr HI H2SO4 HClO4 HNO3
LiOH NaOH KOH Ca(OH)2 Sr(OH)2 Ba(OH)2
pH = -log[H+]
pOH = -log[OH-]
Low pH and high pOH
Low pOH and high pH
H+
OH-
H3O+ (can be used interchangeably with H+)
When acids and bases ionize - fall apart - in solution to form electrolyte solutions
Chemicals that change color in the presence of acids or bases.
An indicator that is used to determine if a solution is acidic or basic. Red litmus turns blue for bases, while blue litmus turns red for acids.
Acid contains H and dissociates to produce H+ ions in aqueous solution, while a base contains OH and dissociates to produce OH- ions in aqueous solution.
An acid is defined as a hydrogen-ion donor and a base is a hydrogen-ion acceptor.
The species produced when a base accepts a hydrogen ion to form an acid.
The species produced when an acid donates a hydrogen ion to form a base.
Two substances related to each other by the donating and accepting of a single H+ ion.
A substance which can behave as either a B/L acid or a B/L base, depending on the circumstances.
An acid that has only one acidic H+ ion
An acid that has two or more acidic H+ ions.
A polyprotic acid that has two acidic H+ ions. An example is H2SO4.
A polyprotic acid that has three acidic H+ ions. An example is H3PO4.