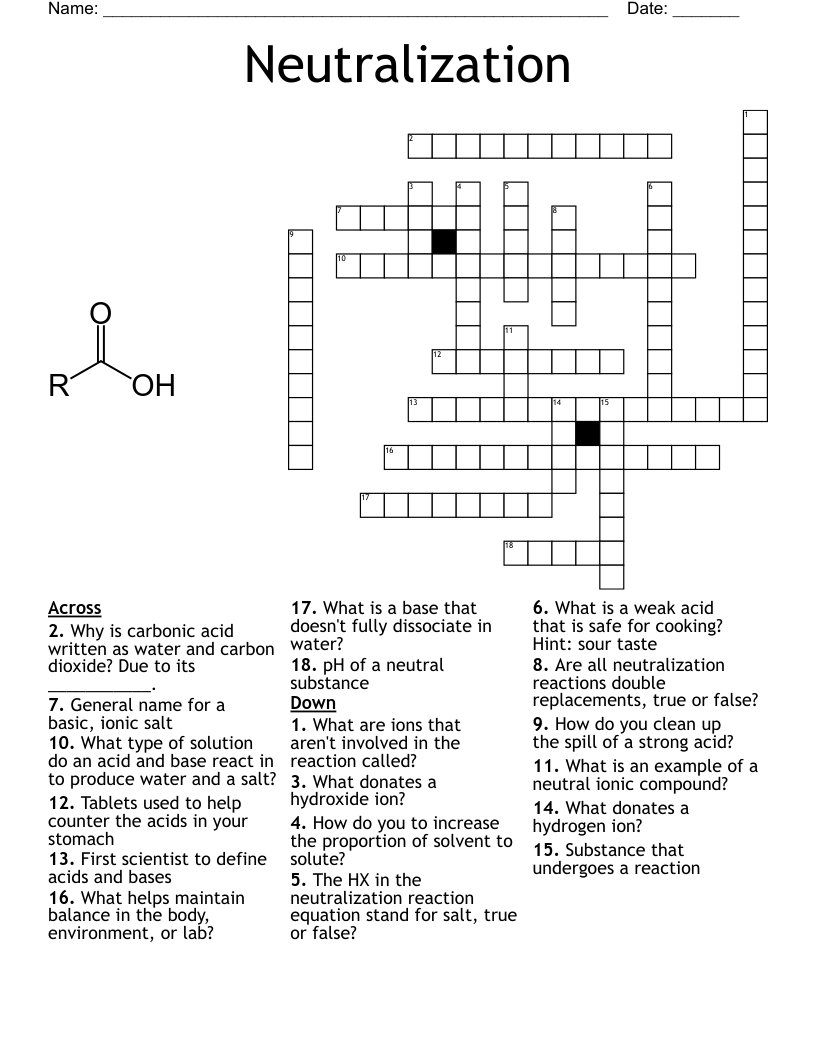
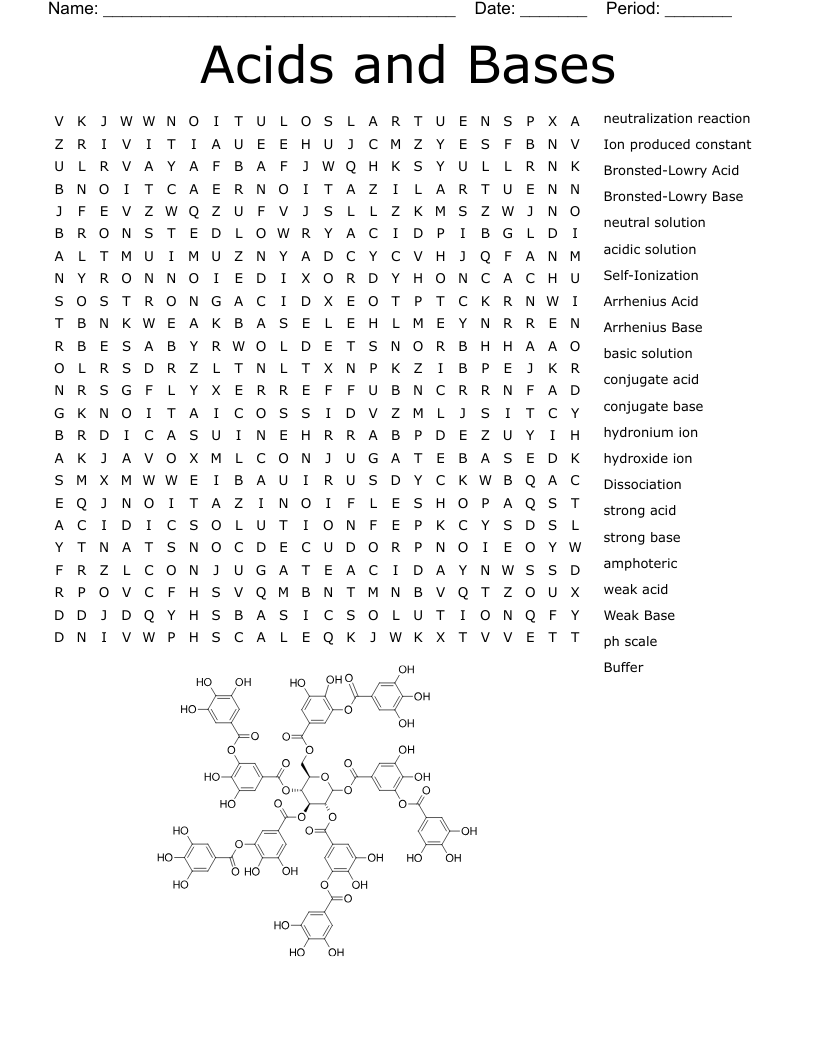
Bases And Acids Part 1 Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 27 clues. Answers range from 4 to 22 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
Produce OH- ions in water Taste bitter, chalky Are electrolytes Feel soapy, slippery React with acids to form salts and water pH greater than 7 Turns red litmus paper to blue
Produce H+ (as H3O+) ions in water (the hydronium ion is a hydrogen ion attached to a water molecule) Taste sour Corrode metals Electrolytes React with bases to form a salt and water pH is less than 7 Turns blue litmus paper to red
Any acid that contains hydrogen and an oxyanion.
An acid which contains hydrogen and one other element. Does not contain oxygen.
A reaction when salt completely dissociates in water, and it's anion or cation react with the water to produce hydroxide ions or hydronium ions that affect the pH of the solution.
An ionic compound made from the cation from a base, and an anion from an acid
A reaction in which an acid and a base in an aqueous solution react to produce a salt and water.
A polyprotic acid that has three acidic H+ ions. An example is H3PO4.
A polyprotic acid that has two acidic H+ ions. An example is H2SO4.
An acid that has two or more acidic H+ ions.
An acid that has only one acidic H+ ion.
A substance which can behave as either a B/L acid or a B/L base, depending on the circumstances. Water is the prototypical amphoteric substance.
Two substances related to each other by the donating and accepting of a single H+ ion.
The species produced when an acid donates a hydrogen ion to form a base.
The species produced when a base accepts a hydrogen ion to form an acid.
An acid is defined as a hydrogen-ion donor and a base is a hydrogen-ion acceptor.
Acid contains H and dissociates to produce H+ ions in aqueous solution, while a base contains OH and dissociates to produce OH- ions in aqueous solution.
An indicator that is used to determine if a solution is acidic or basic. Red litmus turns blue for bases, while blue litmus turns red for acids.
Chemicals that change color in the presence of acids or bases.
When acids and bases ionize - fall apart - in solution to form electrolyte solutions
H3O+ (can be used interchangeably with H+)
OH-
H+
Low pOH and high pH
Low pH and high pOH
pOH = -log[OH-]
pH = -log[H+]