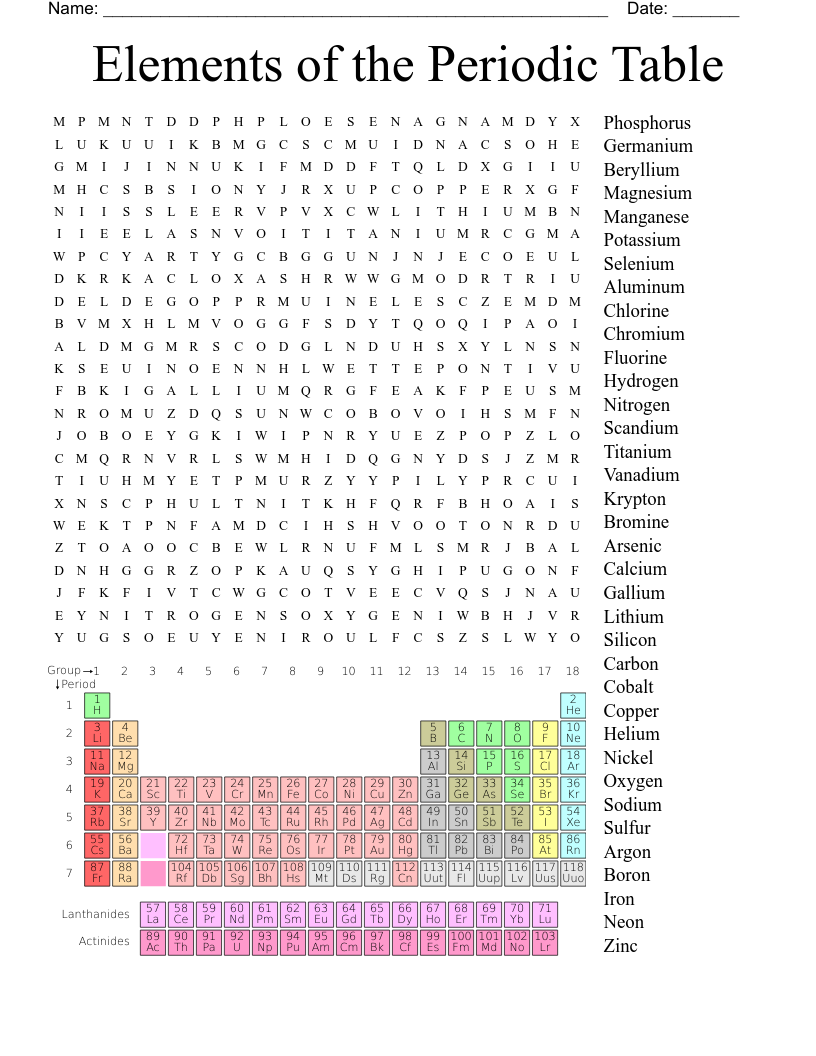
Periodic Table Crossword
This printable crossword puzzle on the topic of Elements & Periodic Table has 20 clues. Answers range from 3 to 21 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
that when the element are arranged by increasing atomic number, there is a periodic repetition of their properties.
a vertical column of elements in the periodic table arranged in order of increasing atomic number; also called a family.
A horizontal row of elements in the modern periodic table.
elements from group 1, 2, and 13-18 in the modern periodic table, possessing a wide range of chemical and physical properties.
elements in groups 3-12 of the modern periodic table and are further divided into transition metals and inner transition metals
an element that is solid at room temperature, a good conductor of heat and electricity, and generally is shiny; most metals are ductile and malleable.
group 1 elements, except for hydrogen, they are reactive and usually exist as compounds with other elements.
Group 2 elements in the modern periodic table and are highly reactive.
an element in group 3-12 that is contained in the d-block of the periodic table and, with some exceptions, is characterized by a filled outermost s orbital of energy level n, and filled or partially filled d orbitals of energy level n-1.
a type of group B element that is contained in the f- block of the periodic table and is characterized by a filled outermost orbital, and filled or partially filled 4f and 5f orbitals.
in the periodic table, the f-block elements from period 6 that follow the element lanthanum.
n the periodic table, the f-block elements from period 7 that follow the element actinium.
elements that are generally gases or dull, brittle solids that are poor conductors of heat and electricity.
a highly reactive group 17 element.
an extremely unreactive group 18 element
an element that has physical and chemical properties of both metals and nonmetals
an atom or bonded group of atoms with a positive and negative charge
the energy required to remove an electron from a gaseous atom; generally increases in moving from left-to-right across a period and decreases in moving down a group.
states that atoms lose, gain, or share electrons in order to acquire the stable electron configuration of a noble gas
indicates the relative ability of an element’s atoms to attract electrons in a chemical bonds.