Warning image from ClipSafari
Acids & Bases Crossword
This printable crossword puzzle on the topic of Chemical Bonding & Reactions has 15 clues. Answers range from 2 to 14 letters long. This crossword is also available to download as a Microsoft Word document or a PDF.
Description
A substance that dissolves in water and produces hydrogen ions (H+), according to the Arrhenius theory. All acids are hydrogen ion donors, according to the Brønsted–Lowry theory.
Substances that can act as either an acid or a base in water.
A substance that dissolves in water and produces hydroxide ions (OH-) according to the Arrhenius theory. All bases are hydrogen ion acceptors, according to the Brønsted–Lowry theory.
A solution of a weak acid and its conjugate base or a weak base and its conjugate acid that maintains the pH by neutralizing added acid or base.
The separation of an acid or a base into ions in water
The point at which an indicator changes color. For the indicator phenolphthalein, the color change occurs when the number of moles of OH- is equal to the number of moles of H3O+ in the sample.
A substance added to a titration sample that changes color when the pH of the solution changes.
The term that describes a solution with equal concentrations of [H3O+] and [OH-].
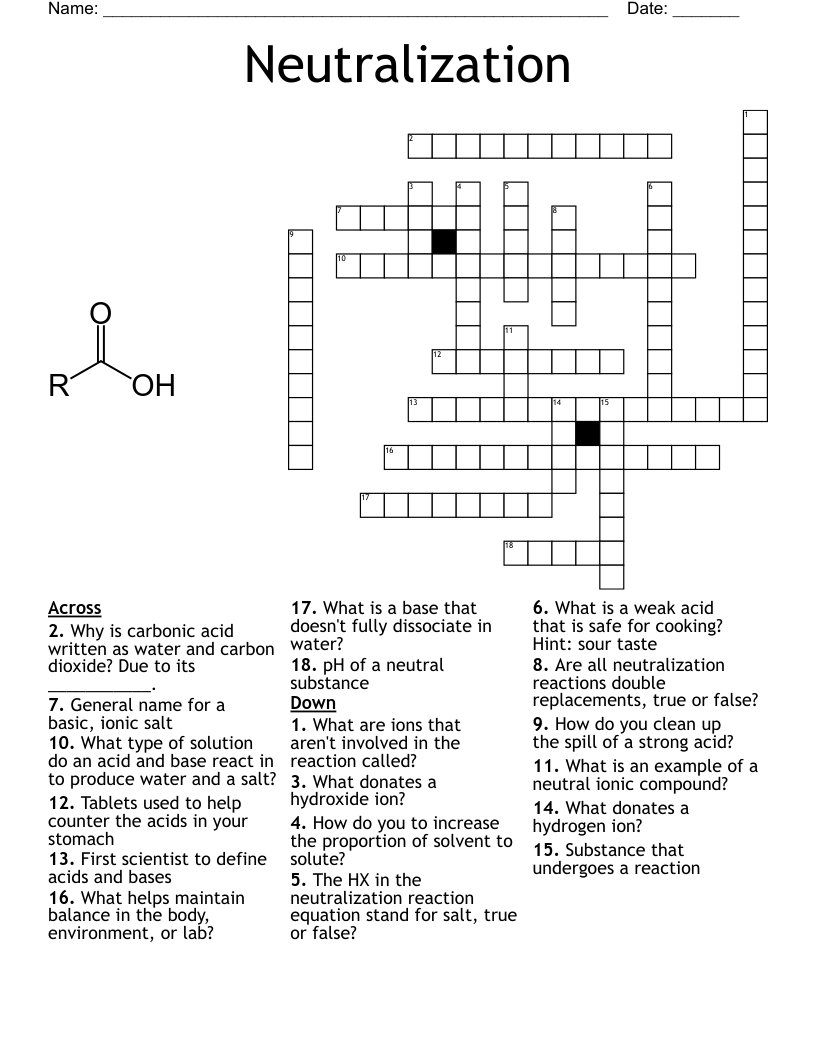
A reaction between an acid and a base to form water and a salt
A measure of the [H3O+] in a solution; pH = -log[H3O+].
A measure of the [OH-] in a solution; pOH = -log[OH-].
An ionic compound that contains a metal ion or NH4 + and a nonmetal or polyatomic ion other than OH-.
An acid that completely dissociates in water.
The addition of base to an acid sample to determine the concentration of the acid.
A base that is a poor acceptor of H+ and produces only a small number of ions in water.
Warning image from ClipSafari